IPO News | Mindray Healthcare (300760.SZ) Submits Hong Kong Stock Exchange Ranks 23rd Among the World's Top Medical Device Companies
The Zhitong Finance App learned that, according to the Hong Kong Stock Exchange's disclosure on November 10, Shenzhen Mindray Biomedical Electronics Co., Ltd. (hereinafter: Mindray Healthcare, 300760.SZ) submitted a statement to the main board of the Hong Kong Stock Exchange, with Huatai International and J.P. Morgan Chase as co-sponsors.

According to the prospectus, the company is a world-leading, innovation-driven world-class medical device company and a leader in medical digital intelligence. According to Frost & Sullivan, in the Chinese medical device industry, the company's market capitalization in 2024 exceeds that of the major domestic companies (2nd to 5th place) combined, and the industrial revenue is more than double that of the second largest. The company has ranked 23rd among the world's top medical device companies, and the ranking continues to rise. It is the only Chinese company among the top 30 medical device companies in the world, and it is also one of the fastest companies from establishment to rise to the list.
The company adheres to a strategic layout and has formed a complete matrix covering multiple products and production lines. According to Frost & Sullivan, the company is the only company among the world's top medical device companies covering multiple diagnosis and treatment scenarios from emergency, operating rooms, intensive care units, general wards to surgery, cardiology, laboratory medicine, and ultrasound. According to Frost & Sullivan's data, in terms of revenue in 2024, the company is in a leading position in the industry in many fields. The market share for 6 categories of products is already in the top 3 in the world, and the market share for 9 categories of products is number one in China.
The company strategically lays out multiple products and multiple production lines. It is the only top medical device company spanning multiple production lines such as in vitro diagnosis, life information and support, medical imaging, minimally invasive surgery, and minimally invasive intervention among the top 30 device companies in the world. The product matrix covers equipment, reagents, consumables and digital intelligence solutions, forming a comprehensive and synergistic product and service system. Based on this, the company is accelerating the strategic transformation of its business layout and comprehensively moving towards a sustainable growth model with flow-based business as the core driving force. Focusing on the core ecosystem of equipment+IT+AI, the company continues to expand its flow-based business represented by reagents, high-value consumables and digital services, and is building new growth momentum characterized by high repurchase frequency and strong customer stickiness. While maintaining its leading edge in the equipment business, the company is actively deepening its strategic layout in emerging fields such as minimally invasive surgery and minimally invasive intervention, improving R&D, registration, production and clinical promotion capabilities through a combination of endogenous and epitaxial methods. Following the direction of minimally invasive business, the company will continue to expand new categories of consumables and clinical application scenarios, promote the upgrading of the Group's business structure from disposable equipment sales to a sustainable, repurchase revenue model, and show more stable and broad development prospects.
The company is a leader in the field of medical digital intelligence. With the company's extensive global installed base and long-term partnerships with leading medical institutions, the company has accumulated and digested a large amount of high-quality data. This allows the company to have the world's largest hardware ecosystem and the most valuable data resources, providing a unique advantage for the company's device+IT+AI ecosystem. Through close cooperation with clinicians, the company continues to deepen its understanding of real practices in multiple departments and scenarios, which in turn promotes the continuous upgrading of Mindray's digital healthcare ecosystem. At the same time, the company successfully released the Qiyuan Model for Critical Care Medicine, which is the world's first major model of intensive medicine to actually achieve clinical implementation. It also recently released the Qiyuan Perioperative Medical Model, the Qiyuan Medical Technology Model, the Qiyuan Obstetrics and Gynecology Model, and the Qiyuan Breast Model. In line with the rapid development of artificial intelligence and medical digital intelligence, the company is accelerating towards becoming a world-leading medical device company.
Financial data
Revenue:
From 2022 to 2024 and the six months ended June 30, 2025, the company's revenue was approximately RMB 30.366 billion, RMB 34.932 billion, RMB 36.726 billion and RMB 16.743 billion, respectively.
Profit:
From 2022 to 2024 and for the six months ended June 30, 2025, the company's profits were approximately RMB 9.611 billion, RMB 11.780 billion, RMB 11.740 billion, and RMB 5.233 billion, respectively.
Gross profit margin:
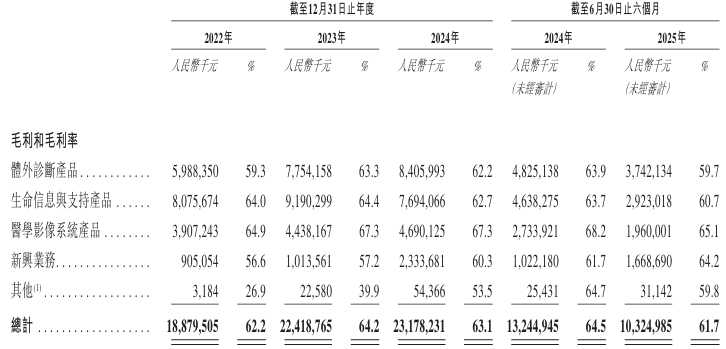
From 2022 to 2024 and the six months ended June 30, 2025, the company's gross margins were 62.2%, 64.2%, 63.1%, and 61.7%, respectively.

Industry Overview
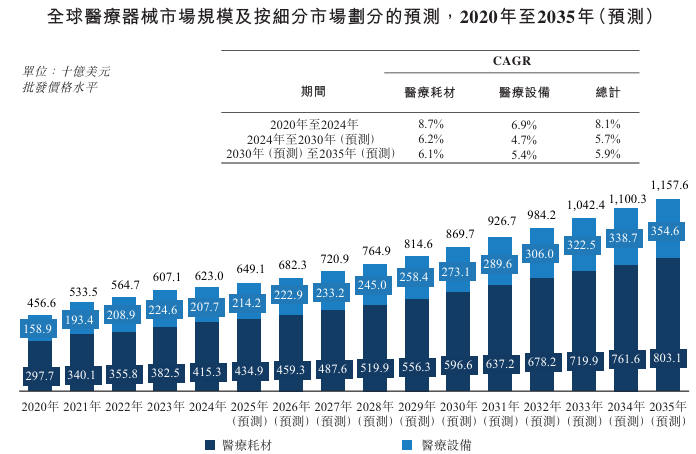
The global medical device market grew from US$456.6 billion in 2020 to US$623 billion in 2024, with a CAGR of 8.1% from 2020 to 2024. Driven by basic healthcare needs and continued technological advances, the market is expected to maintain a steady growth momentum, reaching US$869.7 billion in 2030, with a CAGR of 5.7% from 2024 to 2030; further increasing to US$1,157.6 billion by 2035, and a CAGR of 5.9% from 2030 to 2035. This continued expansion reflects the combined impact of demographic changes, increased healthcare awareness, and the acceleration of the digitization of global health services.
Among them, the Chinese medical device market grew from RMB 729.8 billion to RMB 941.7 billion from 2020 to 2024, with a CAGR of 6.6%. The market size is expected to reach RMB 1,326 billion by 2030, with a CAGR of 5.9% from 2024 to 2030; further increasing to RMB 1813.4 billion in 2035, and a CAGR of 6.5% from 2030 to 2035.

The continued growth of the global medical device industry is driven by structural healthcare needs, technology convergence, supportive policy frameworks, and evolving business models. These drivers are reshaping how healthcare is delivered and accelerating the digital and intelligent transformation of the industry.
As the world's second-largest medical device market, China has maintained strong structural growth. This is due to the expansion of primary care infrastructure, the spread of chronic disease management solutions, and supportive government policies to promote device renewal and independent innovation. The integrated business model of equipment+consumables/reagents+services continues to enhance customer loyalty and product stickiness. Although volume procurement policies are putting downward pressure on unit prices, the resulting market size growth and industry consolidation are expected to support the long-term sustainable growth of leading manufacturers.
Policies are a key factor in shaping the medical device industry, affecting innovation, market access, and pricing. Over the past five years, China has implemented a series of strong policies for the medical device industry around encouraging innovation and improving accessibility with two-wheel drive. In order to optimize the review and approval process, establish a fast track for innovative devices, and support R&D activities, relevant policies have been introduced one after another to promote the development of the medical device industry in the direction of high-end intelligence. Health insurance reimbursement reforms, government-led device renewal programs, and the promotion of a hierarchical diagnosis and treatment system have accelerated the market entry and widespread application of innovative products, benefiting a wider range of patients.
Board Information
The board of directors consists of 12 directors, including 5 executive directors and non-executive directors, and 7 independent non-executive directors.



Shareholding structure
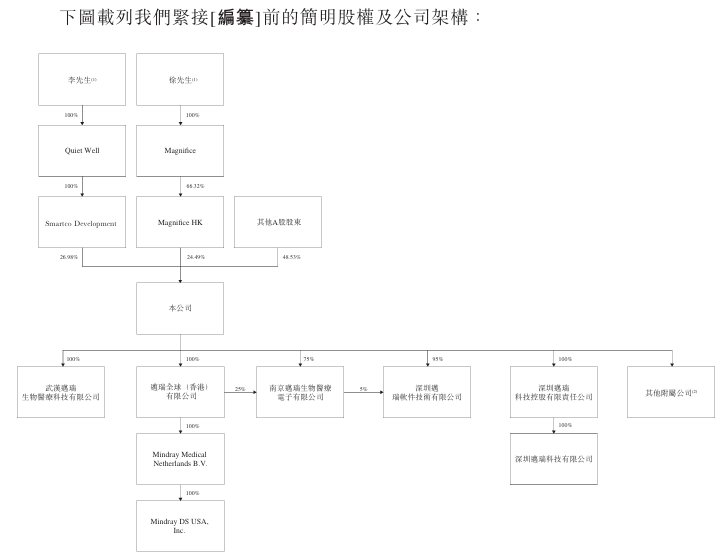
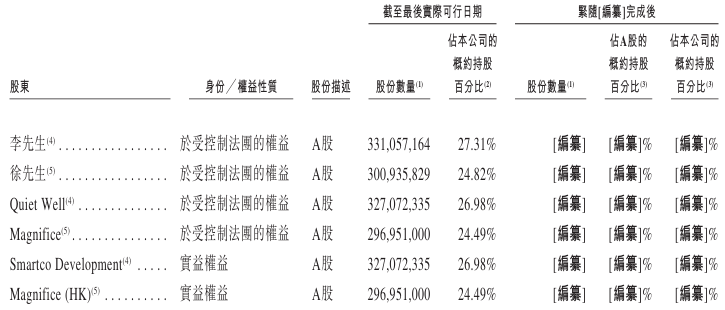
As of the last practical date (November 3, 2025), SmartCodeDevelopment held 327,072,335 A-shares. SmartCodeDevelopment is wholly owned by QuietWell, while QuietWell is wholly owned by Mr. Lee. Therefore, under the Securities and Futures Ordinance, Mr Lee and QuietWell are each deemed to have an interest in shares held by SmartCodeDevelopment. Furthermore, as of the last practical date, Shenzhen Ruifu Management Consulting Partnership (Limited Partnership) (Shenzhen Ruifu) held 2,747,306 A shares, while Shenzhen Ruilong Consulting Service Partnership (Limited Partnership) (Shenzhen Ruilong) held 1,237,523 A shares. Since Mr. Li holds limited partnership interests of Shenzhen Ruifu and Shenzhen Ruilong each, Mr. Li is also considered to have interests in A-shares held by Shenzhen Ruifu and Shenzhen Ruilong respectively according to the Securities and Futures Regulations.
As of the last practical date, Magnifice (HK) held 296,951,000 A-shares. Magnifice (HK) is 66.32% owned by Magnifice, while Magnifice is wholly owned by Mr. Xu and 33.68% by SimpleWellLimited, while SimpleWellLimited is controlled by Ms Jane Yao through SimpleWell (BVI) Holding Limited. Therefore, under the Securities and Futures Ordinance, Mr Tsui, Magnifice, SimpleWell Limited, SimpleWell (BVI) HoldingLimited and Ms Jane Yao are all considered to have interests in shares held by MagniFice (HK).
Furthermore, as of the last practical date, Shenzhen Ruifu held 2,747,306 A shares, while Shenzhen Ruilong held 1,237,523 A shares. Since Mr. Xu holds limited partnership interests of Shenzhen Ruifu and Shenzhen Ruilong each, Mr. Xu is also considered to have interests in A-shares held by Shenzhen Ruifu and Shenzhen Ruilong respectively according to the Securities and Futures Regulations. Furthermore, at the last practical date, Magnifice (HK) has pledged 36,100,000 A-shares held by Magnifice (HK) to a number of regulated financial institutions (such as bank branches in China within the list of authorized institutions as defined in the Banking Regulations) as collateral for certain financing provided by such financial institutions to Magnifice (HK).
Intermediary team
Co-sponsors: Huatai Financial Holdings (Hong Kong) Limited, J.P. MorganSecurities (FarEast) Limited
Legal Advisors: DavisPolk & Wardwell, Fangda Law Firm
Co-Sponsors and Compiling Legal Advisors: Linklida Law Firm, Jingtian Gongcheng Law Firm
Reporting accountant and auditor: Ernst & Young
Industry Advisor: Frost & Sullivan (Beijing) Consulting Co., Ltd. Shanghai Branch
Compliance Advisor: Maxi Capital Limited