IPO News | Changfeng Pharmaceutical successfully became the highest-selling inhaled drug category in China through CF017 commercialization
The Zhitong Finance App learned that according to the Hong Kong Stock Exchange disclosure on September 22, Changfeng Pharmaceutical Co., Ltd. (“Changfeng Pharmaceutical” for short) passed the Hong Kong Stock Exchange listing hearing, and CITIC Securities and CMB International were co-sponsors.

According to the prospectus, Changfeng Pharmaceutical was founded in 2007 and focuses on R&D, production and commercialization of inhalation technology and inhaled drugs, focusing on the treatment of respiratory diseases.
As the prevalence of respiratory diseases such as asthma, COPD, and allergic rhinitis continues to rise, the global respiratory drug market is huge. It is estimated that nearly 2.5 billion people around the world currently suffer from chronic respiratory diseases, and this number is expected to increase in the future. In 2024, the global respiratory medicine market is estimated at US$99.9 billion (approximately RMB 729.2 billion) and is expected to reach US$157.2 billion (approximately RMB 1,147.4 billion) by 2033, with a compound annual growth rate of 5.2%.
CF017, an inhaled budesonide suspension for the treatment of bronchial asthma (the most sold inhaled drug category in China), is Changfeng Pharmaceutical's first approved product. After approval in May 2021, CF017 was quickly incorporated into the China Centralized Procurement (VBP) program and achieved market growth. According to Frost & Sullivan, in 2024, CF017 accounted for about 16% of China's budesonide inhaled drug market in 2024 in terms of sales volume.
Since it was approved in May 2021, Changfeng Pharmaceutical's marketing system has enabled CF017 to successfully cover more than 10,000 medical institutions in China. The company's CF018 is the first azetistine fluticasone nasal spray approved for the treatment of moderate to severe allergic rhinitis in China. After being included in the 2023 “National Medical Insurance Drug Catalogue” (National Health Insurance Catalogue), it has penetrated more than 500 hospitals and medical institutions in many provinces.
Over the years, Changfeng Pharmaceutical has built five major technical platforms covering the core aspects of inhalation formulation development, namely particle engineering, device design, product performance evaluation, clinical development and process engineering, and has built a solid foundation in the research pipeline.
Today, Changfeng Pharmaceutical is promoting the global development of more than 20 candidate products in major markets such as China, the US and/or Europe, and emerging markets such as Southeast Asia and South America. In addition to first-stage inhalation products, the company is also driving global innovation and breaking the boundaries of formulation application. The company is exploring novel inhaled formulations, such as liposomes and siRNA, and entering new therapeutic areas, including CNS diseases and anti-infectives. Furthermore, the company is developing new treatments such as endobronchial valve activation (EBV), and developing potential first-of-its-kind or first-of-its-kind treatments for diseases such as IPF and PAH that seriously affect patients' lives.
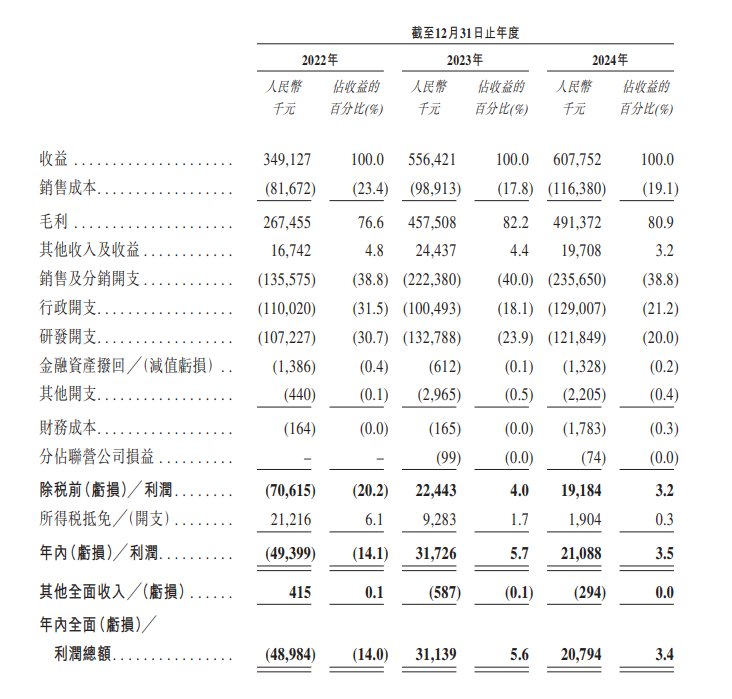
On the financial side, thanks to the successful commercialization of CF017, Changfeng Pharmaceutical's revenue increased from 349 million yuan (RMB, same below) in 2022 to 608 million yuan in 2024, with a compound annual growth rate of 31.9%; in 2024, it achieved gross profit of 491 million yuan, an increase of about 7.2% over the previous year. Furthermore, the company benefits from operating efficiency brought about by economies of scale and will continue to actively control administrative expenses. It is expected that the ratio of administrative expenses to revenue will continue to decline as the business expands.