Tylomac's Innovation Board IPO “inquired” and the core product, Stedutazumab injection, was approved for listing in China in February this year
The Zhitong Finance App learned that on August 11, Zhuhai Tainomaibo Pharmaceutical Co., Ltd. (abbreviation: Tainomaibo) applied for listing on the Shanghai Stock Exchange Science and Technology Innovation Board and changed its review status to “Inquired”. Huatai United Securities is its sponsor and plans to raise 1500 billion yuan.
According to the prospectus, Tylomaibo is an innovative biopharmaceutical company dedicated to the global market and dedicated to alternative therapies for blood products. After years of accumulation, the company has built a number of core technology platforms such as “HitMAb®, a comprehensive technology platform for high-throughput all-human monoclonal antibody research and development,” “high efficiency antibody expression CHO-GS cell platform,” “process development platform,” and “bioanalysis platform” with independent intellectual property rights, and has continuously cultivated and perfected innovative drug technology capabilities covering drug discovery, engineered cell line construction, process and quality development, pharmacological research, toxicology research, pharmacology research, clinical development, and large-scale production in line with international GMP standards.
The company's core product, stedutazumab injection (i.e., “recombinant anti-tetanus toxin all-human monoclonal antibody TNM002”, trade name: cintetal®, hereinafter referred to as “stedutazumab injection” or “TNM002”) was approved for marketing in China in February 2025. It is the world's first first-in-class (first-in-class) recombinant anti-tetanus monoclonal antibody drug. It is also the first domestically produced innovation in the field of anti-infection to be recognized as a breakthrough therapeutic drug (Biopharmaceuticals), has been included in the Fast Track (Fast Track) qualification by the US FDA, and has world-leading revolutionary significance in the field of tetanus prevention.
Another core product of the company, “recombinant anti-respiratory syncytial virus all-human monoclonal antibody TNM001” (hereinafter referred to as “TNM001”), is potentially the third in the world and the first long-term anti-RSV monoclonal drug for preventive use in healthy and high-risk infants. As of the date of signing of this prospectus, TNM001 is undergoing phase III clinical trials.
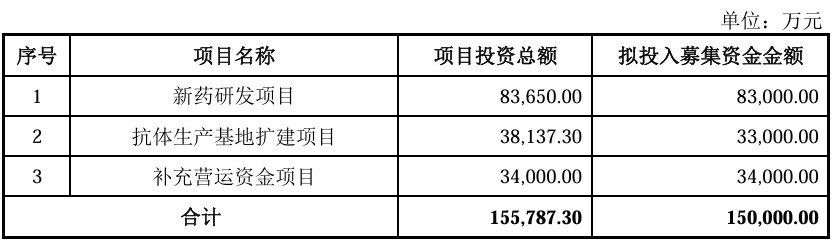
After deducting costs such as issuance, the actual capital raised in this issue will be used for projects related to the company's main business and working capital required for the development of the main business. The specific investment projects are as follows:
On the financial side, in 2022, 2024, and January-March 2025, Tylomaibo's revenue was approximately RMB 4.3398 million, RMB 15.559 million and RMB 169,300, respectively; in 2022, 2023, 2024, and January-March 2025, the company achieved net profit of approximately -429 million yuan, -446 million yuan, -515 million yuan and -177 million yuan, respectively.
