Heyu-B (02256) mid-report view: using “hematopoietic advantage” to raise the margin of safety, and rely on hard core innovation to enhance allocation value
On July 29 of this year, the highest intraday share price of WAYU-B (02256) reached HK$11.74, setting a record high in the company's stock price in the past 4 years. This has also brought the biggest increase in Hewin's stock price from the beginning of the year to date to 155.22%.
While rising stock prices have boosted investment returns for investors in the secondary market, Heyi also continues to use “real money” to give back to investors through cancellation stock repurchases. In March of this year, the board of directors of Heyu once again approved the use of HK$200 million to repurchase shares in the market. As of June 30 this year, the company has repurchased a total of 9.545 million shares, totaling HK$75.3 million.
In fact, since this year, the innovative drug sector of the Hong Kong stock market has gone through a wave of explosive sales. Investors can easily see from Heyu's recently disclosed 2025 mid-term earnings report that with the continuous fulfillment of innovative R&D and international BD expectations, the company successfully achieved continuous large-scale innovation benefits and profits, and generated abundant cash flow, providing a considerable financial guarantee for the company's medium- to long-term innovative research and development, and further improving the company's “innovation-profit” cycle. The rising stock price in the first half of this year was reflected in the market's high recognition of the company's value.
Large-scale profit expectations continue to be fulfilled
In the first half of this year, Horizon was able to achieve continuous fulfillment of large-scale profit expectations. The key carrier is pimitinib, the core product that continues to release commercial value.
As the first CSF-1R inhibitor independently developed by China to enter the global phase III TGCT clinical trial, pimitinib has now been recognized as a breakthrough therapy by regulators in China, the US, and Europe. As a potential global BIC drug on the CSF-1R inhibitor circuit, pimitinib not only has outstanding TGCT indications, but also has more room for imagination in various indications such as cGVHD.
Precisely because the drug's “billion-dollar molecule” potential is outstanding, the company reached a “high down payment, high milestone, and high share” BD deal with Merck, a well-known multinational pharmaceutical company, at the end of 2023, and received a down payment of 70 million US dollars at the beginning of '24.
The Zhitong Finance App learned that in November of last year, the positive top-line results of the global Phase III Pimitinib study were released. Meanwhile, in the first half of this year, after receiving approval from the Chinese NMPA in May for priority review, pimitinib's NDA application for the treatment of TGCT was approved by the NMPA in June. At present, there is a high degree of certainty that the drug has been approved domestically.
In addition, pimitinib has also been granted BTD by the Chinese NMPA and the US FDA, respectively, and PRIME certification by the European Medicines Agency ([EMA)] for the treatment of inoperable TGCT patients. At the same time, it has also obtained Fast Track Accreditation (FTD) from the US FDA and Orphan Drug Eligibility (ODD) from the EMA.
Pimitinib has been certified by major authorities around the world, which also indicates that commercialization is about to begin. On April 1 of this year, Goodwill officially announced that Merck has exercised the global commercialization option of pimitinib (ABSK021) in accordance with the licensing agreement signed by the two parties in December 2023, at a cost of 85 million US dollars. This was also one of the direct factors driving Wakeup's continued profit in the first half of this year.
According to financial reports, the company achieved total revenue (including revenue and other revenue and revenue) of 657 million yuan (RMB, same unit), an increase of 20% year on year; at the same time, the company recorded current net profit of 328 million yuan to mother, an increase of 59% over the previous year, and the current adjusted net profit reached 336 million yuan.

In fact, in addition to Merck's current exercise of rights payment, the BD cooperation between the two parties has brought over 150 million US dollars in cash to Hewin. According to the previous agreement, Kazuyo will also receive R&D milestone payments and sales milestone payments from Merck in the future. The total potential payment may be as high as 606 million US dollars, not including the double-digit net sales share. And these funds will all become the company's subsequent continuous cash flow, bringing clear profit to the company.
It is easy to see that strong innovative R&D capabilities and global BD capabilities have gradually turned Hewin's clinical R&D into fruitful commercialization results, thereby continuing to promote the company's innovative research and development, shortening the development cycle, and paving the way for the smooth implementation of many major FIC/BIC varieties in the future.
Improve the closed loop of innovation and consolidate the foundation for development
There is no doubt that the strong commercialization expectations of the core product pimitinib are an important reason why the valuation of Heyu has achieved leaps and bounds this year, but the gradual improvement of the “innovation-profit” closed loop established by core innovative research and development cannot be ignored.
Financial reports show that in the first half of this year, Heyu confirmed that R&D investment reached 228 million yuan, with stable revenue and cash flow growth. The strong support from the innovative R&D side brought to the company, consolidated the company's foundation for exploring the global differentiated innovative drug market, and accelerated the company's advance to the first tier of global Biopharma.
In recent years, Heyu Pharmaceutical has focused on precise tumor treatment and immunotherapy, covering popular targets such as EGFR, FGFR, CSF-1R, and PMRT5, and has established 22 differentiated innovative R&D pipelines with global competitiveness. Many of the innovative drugs under development have the potential to be “best-in-class” or “world first”.
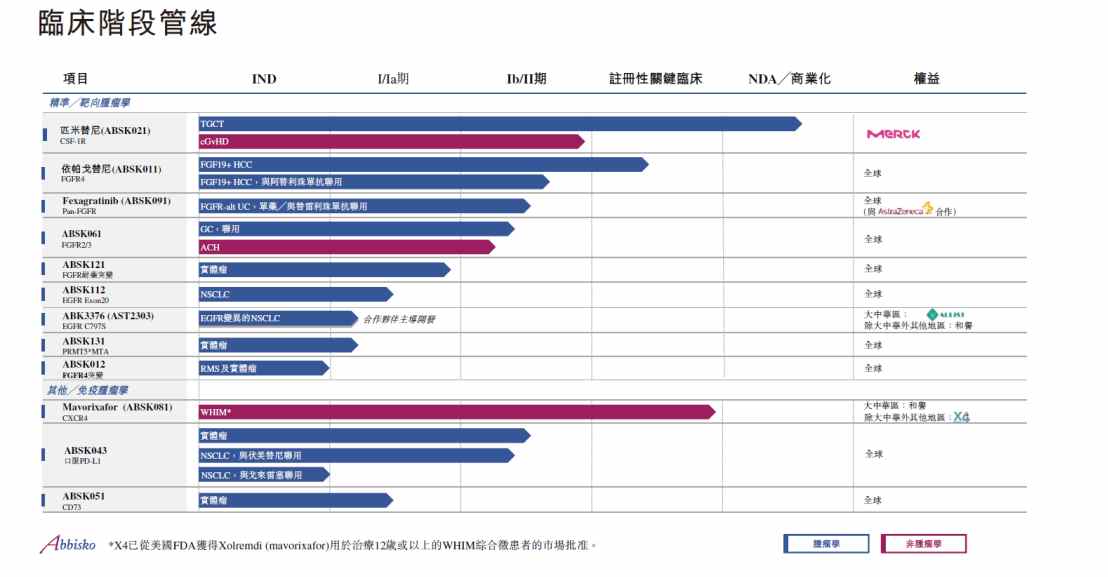

And ipagotinib (ABSK011), which is expected to become a global FIC drug similar to FGFR4 inhibitors, is certainly one of the best.
On May 26 this year, Heyu officially announced that ipagotinib (ABSK011) has been included as a breakthrough treatment for patients with advanced FGF19 overexpressed hepatocellular carcinoma who have previously been treated with immune checkpoint inhibitors (ICI) and multi-target tyrosine kinase inhibitors (mTKI). After pimicotinib (pimicotinib/absk021), this is the second major variety of Heyuan to be included as a breakthrough therapy.
In fact, as the world's first small molecule inhibitor targeting abnormal activation of the FGF19/FGFR4 signaling pathway, ABSK-011 has received widespread attention from the global industry and market due to its remarkable efficacy and greater compliance and economic advantages compared to other FGFR4 targeted drugs since its debut at the 2023 ESMO Annual Conference.
From a market perspective, according to GlobalData's forecast, the global liver cancer market will be around US$5.3 billion by 2029, of which immunotherapy accounts for about 72.2% of the market share, reaching US$3.8 billion. Referring to the previously marketed small-molecule kinase inhibitor sorafenib, its ORR for liver cancer indications is less than 20%. Even so, its global sales in 2021 still exceeded 500 million US dollars, reflecting the huge unmet treatment demand in the current market.
However, the key to Heyu Pharmaceutical's ascendancy was innovation and breakthroughs in molecular design. According to reports, Heyu's R&D team screened out highly selective compounds through thousands of experiments, and successfully avoided the side effects of off-target FGFR1/2/3. Therefore, as Heyu's second major variety to be included as a breakthrough therapy, ipagotinib is highly definitive, and with its leading position in the competitive landscape, it has now “overtaken a corner” at the R&D level, and is expected to become the first breakthrough drug to treat patients with FGF19 overexpressing HCC.
In addition to FGFR targets, there are a number of high-quality pipelines in the Heyu Innovation Pipeline that are in the early clinical and pre-clinical stages. In the first half of this year, Heyu once again brought 4 of the latest pre-clinical research results to this year's AACR conference and published the latest preclinical research results of its self-developed EGFR exon20ins inhibitor ABSK112, PRMT5*MTA inhibitor ABSK131, and KRAS inhibitors in the form of posters, showing the global academic community its R&D achievements in the field of small molecule innovation.
All of the above reflect Heyu's certainty in global commercialization and differentiated original research innovation. Heyu is expected to open up more room for valuation imagination through multiple potential FIC/BIC products within the pipeline.
Referring to the current valuation of innovative pharmaceutical companies in the commercialization stage of Hong Kong stocks, Yasheng Pharmaceutical's PS valuation reached 26.65 times, and Kangfang Biotech reached 60.53 times. In contrast, Heyu is currently in the early stages of commercialization, holding rich cash of 2.3 billion yuan and being able to continue to return shareholders through stock repurchases. Currently, the company's PS valuation is only 14.01 times, and there is still huge room for growth. It is in line with the current market's long-term investment logic for scarce innovative drug brands with high returns, and is worthy of long-term attention from investors.